Cytotoxicity Tests in Mammalian-, Prokaryotic Cells (Ames Test)
Cell Viability Assay in Mammalian and Bacterial Cells - Principle
Chemical compounds, pharmaceuticals, cosmetic products, natural plant extracts or materials used for implants can have a toxic effect on bacterial or on mammalian cells. There are different mechanisms of toxicity, i.e., loss of membrane integrity, lysosomal toxicity, inhibition of protein synthesis or interference in metabolism which can be detected by various technologies.
Not only for the development of pharmaceuticals, cosmetics, plant extracts or chemicals and implants, but also for the environment it is important to discriminate between no toxicity, negligible and strong toxicity. For example, compounds with promising biological activity and negligible cytotoxicity should not be ruled out, because “The dose makes the poison", i.e., all things are poison, and nothing is without poison, the dosage alone makes it so a thing is not a poison (Paracelsus, 1493–1541).
Various bioassays and several different eukaryotic and prokaryotic cell lines are used to assess cytotoxicity. Cytotoxicity assays detect cellular or metabolic changes associated with viable or nonviable cells. Different assays can detect different mechanisms in cells, such as biochemical activities in the electron transport chain, protein synthesis, lysosomal activity, or loss of membrane integrity, which are indicative signs of living cells.
For example, tetrazolium salts like XTT or other redox dies are a valuable tool to assess quantitatively viable cells, since dyes are only reduced by metabolic active cells.
Cytotoxicity in the Ames Test
The Ames test, a bacterial reverse mutation assay detects chemical compounds which induce mutations in the bacterial genome enabling the cells to grow without histidine (Salmonella) or Escherichia coli. Most compounds show a classic dose–response curve if number of reverted bacteria are plotted versus concentration of the test chemical. If cytotoxicity occurs, the dose–response curve shows linear initial slope due to the accumulation of mutations with a subsequent downward curve showing cell dying (cytotoxicity) at higher doses of the test chemical. Lower number of revertants with increasing concentration of test chemical correlates with cytotoxicity.
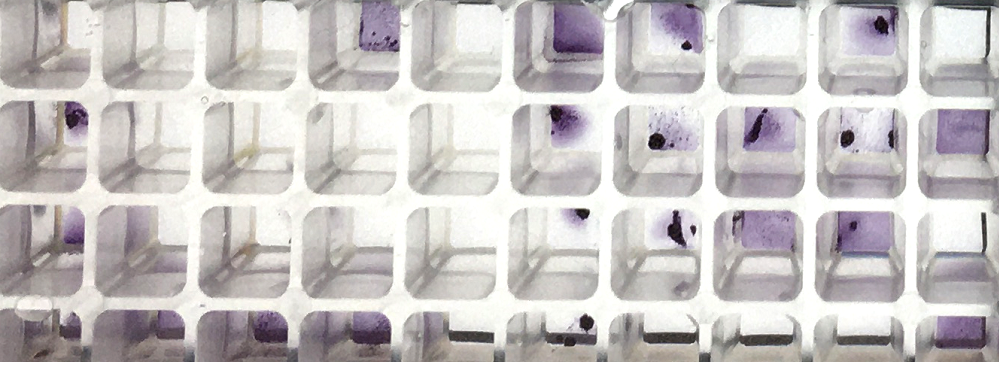
Under cytotoxic or antimicrobial conditions bacteria form so called microcolonies, which can be detected by eye on the agar plate in the Ames test. In the liquid microplate format Ames MPF™ bacterial cells form an alignment of cells which can be observed as lines under the microscope. Cytotoxicity can also be detected by the intensity of the purple color in cytotoxic wells as well as small bubbles of the liquid Ames test Ames MPF™. Strong purple color of the wells of a cytotoxic compound as compared to negative control (unless volatile) is associated with cytotoxicity. Cytotoxicity can also be detected by measuring metabolism in the Ames MPF™ Test. Certain colorants allow kinetic analysis of homogenous dye formation by cells in culture medium. The quantity of dye produced is usually directly proportional to the number of living and respiring cells. Product in development.
 |
Example of cytotoxicity caused by 4-Nitro-Quinoline (4-NQO): purple colored metabolic active cells (upper 48 wells) and same cells with cytotoxic dose of 4-NQO (lower 48 wells), uncolored cells with no metabolic activation. |
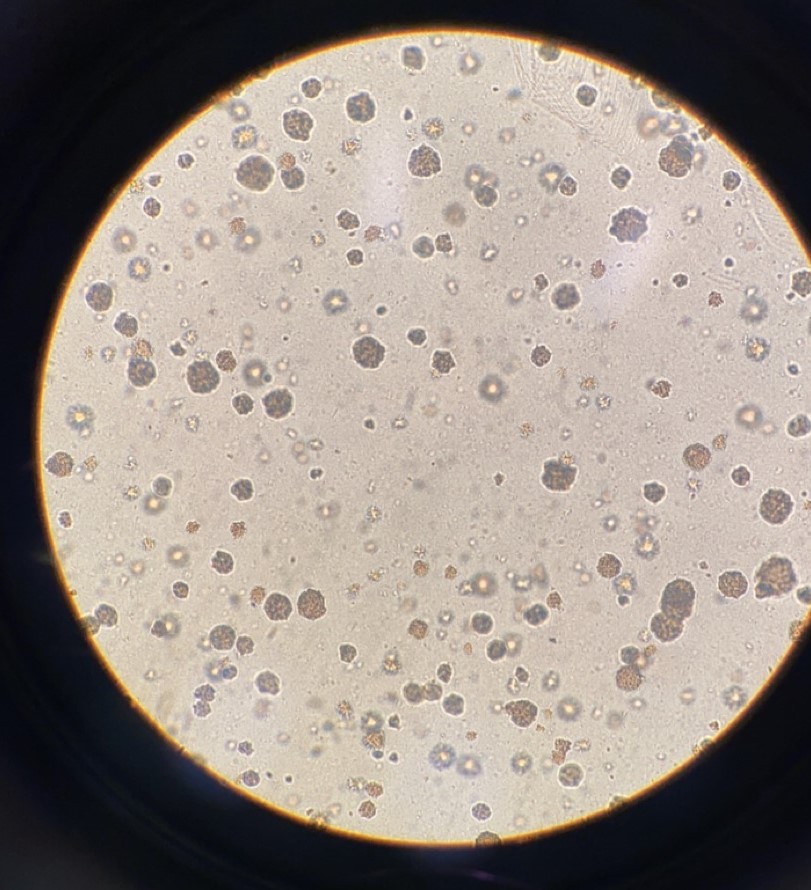 |
Formation of Microcolonies by a cytotoxic compound in the agar based MicoAmes24 Test |
Ames Assay Service Analytics available for cytotoxic compounds, proteins, aminoacids, antibodies, etc., please contact info@xenometrix.ch
Microsomal Rat Liver S9 Fraction
Microsomal S9 fraction of rat liver used in the Ames Test can have a cytotoxic effect on the bacterial cells. S9 mimics the liver metabolism in the in vitro Ames test system. Metabolization of test compounds must be taken into account in all genotoxic studies and environmental safety assessments of chemicals according to OECD TG471, M7 or in vitro mammalian cell micronucleus test OECD TG487.
Cytotoxicity - Description of the Cytotoxicity Assay in Mammalian Cells
The XTT assay is based on the cleavage of the yellow tetrazolium salt XTT to form an orange formazan dye. The formazan dye formed from XTT is soluble in aqueous solutions and can be monitored quantitatively by a microplate spectrophotometer. The quantity of formazan product is directly proportional to the number of living and respiring cells.
Most assays can be automated allowing high throughput processing. Implants or chemicals used in implants have to be tested for cytotoxicity according to ISO 10993.
A usual XTT cell viability assay is shortly described here: Cells are grown in 96 well tissue culture plates and are incubated with one of the above mentioned stain solution and test compound during 4–24 h. After this incubation period, formazan solution is formed, which is spectrophotometrically quantified. An increase in number of living cells results in an increase in the OD.
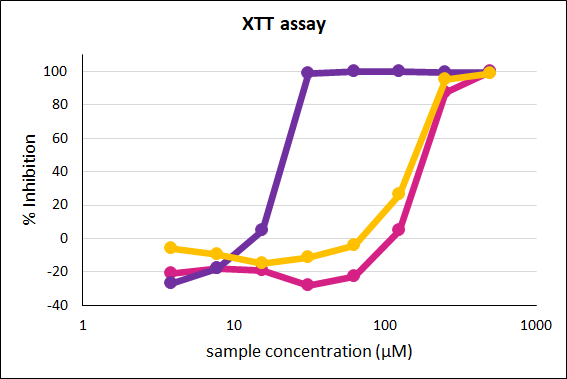 |
Inhibition of formazan production is plotted against the test compound concentration on a percentage scale for three test items. The IC50 value (concentration at which the maximal production is halved) can be determined graphically. |

 Navigation
Navigation