Miniaturized Ames Test
Miniaturized Ames test – Principle
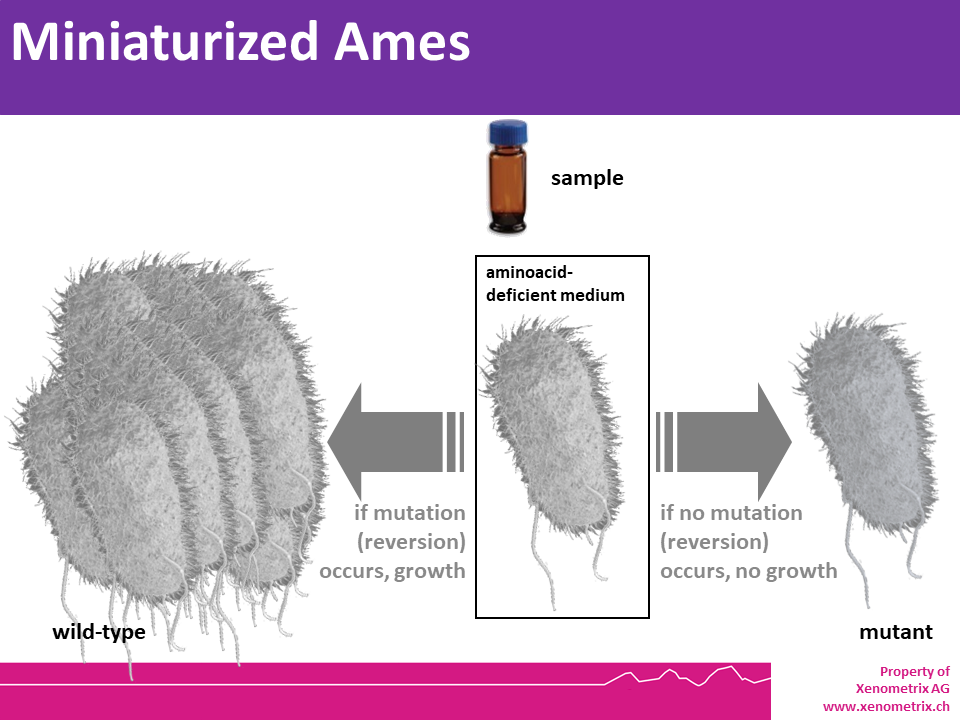
The principle of the miniaturized Ames assays is identical to the agar based Ames test OECD TG 471. Point mutations were made in the histidine (Salmonella typhimurium) or the tryptophan (Escherichia coli) operon, rendering the bacteria incapable of producing the corresponding amino acid. These mutations result in his- or trp- organisms that cannot grow unless histidine or tryptophan is supplied.
The mutagenic potential of a test sample is assessed by exposing these amino acid-requiring organisms to varying concentrations of sample and selecting for the reversion event. Media lacking histidine or tryptophan are used for this selection which allow only those cells that have undergone the reversion to histidine / tryptophan prototrophy to survive and grow. A mutagenic event (causing either base substitutions or frameshift mutations) within the gene may cause a reversion to amino acid prototrophy. These reverted bacteria will then grow in histidine- or tryptophan-deficient media, respectively, whereas non reverted bacteria will not be able to grow. The media during the growth phase can be liquid or agar based.
Different Forms of miniaturized Ames test systems
- Miniaturized Ames test Ames MPF™ based on liquid microplate technology – 55 mg of test compound per 5 strains, +/- S9
- Miniaturized Ames test MicroAmes24 based on 24 well agar plate – 30 mg of test compound per 5 strains, +/- S9
- Ultra-miniaturized Ames test NanoAmes™ based on 25 square well agar plates – 35 μg of test compound per 5 strains, +/- S9, available as service analytics only
Miniaturized Ames test in Agar Plates – Assay Description
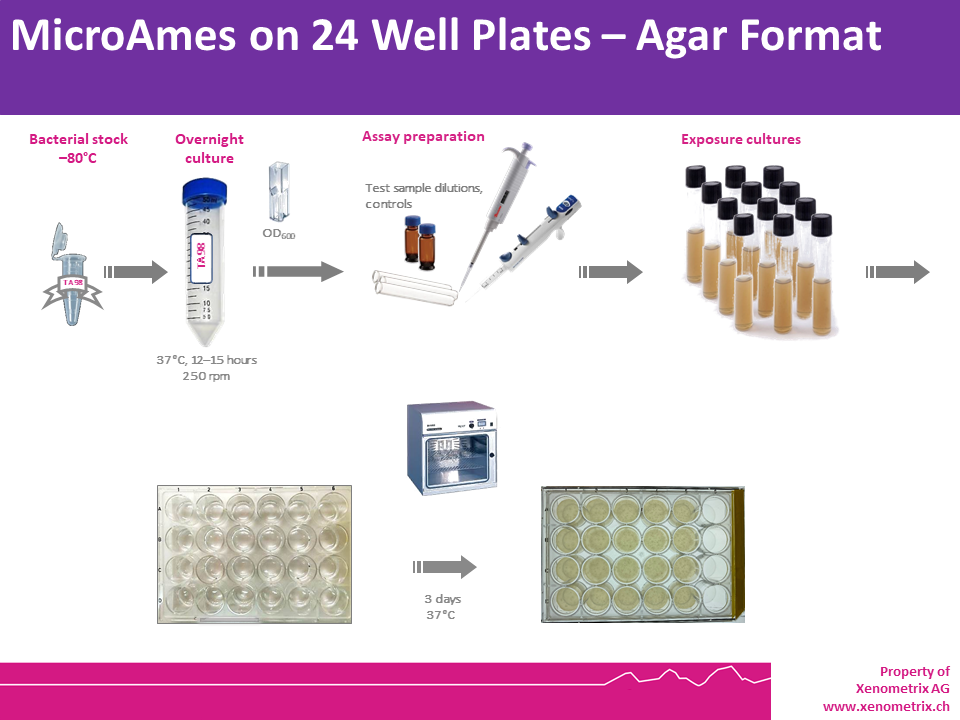
Bacteria are exposed to different and low concentrations of a test sample, as well as a positive and a negative control in a medium containing limited quantities of histidine (S. typhimurium) or tryptophan (E. coli) to support approximately two cell divisions. After exposure (pre-incubation test) or without exposure (plate incorporation test), The cultures in each condition (negative control, test samples and positive controls) are poured on an agar support in multiwell plates (24 well plates) and are incubated. Reducing the surface area (comparatively to a Petri dish), the amount of test sample needed to run the test is dramatically reduced, while keeping similar limits of detection.
Within three days, cells that have undergone reversion to amino acid prototrophy will grow and form colonies, whose counts will be compared to those grown in the solvent (negative) control wells. Each dose is tested in triplicate to allow for statistical analysis of the data.
A dose-dependent and significant increase in the number of revertant colonies upon exposure to test sample relative to the solvent controls indicates that the sample is mutagenic.
The mutagenic potential of samples is assessed directly, the compounds are tested in the absence and presence of metabolic activation, provided by a liver homogenate, a post-mitochondrial supernatant fraction S9, respectively.
More information on MicroAmes24 – Miniaturized 24-well agar plate Ames test
More information on NanoAmes™ – Ultra-Miniaturized 25-well agar plate Ames test – available via Service Analytics
Miniaturized Ames test in liquid 384 Well Microplate Format – Assay Description
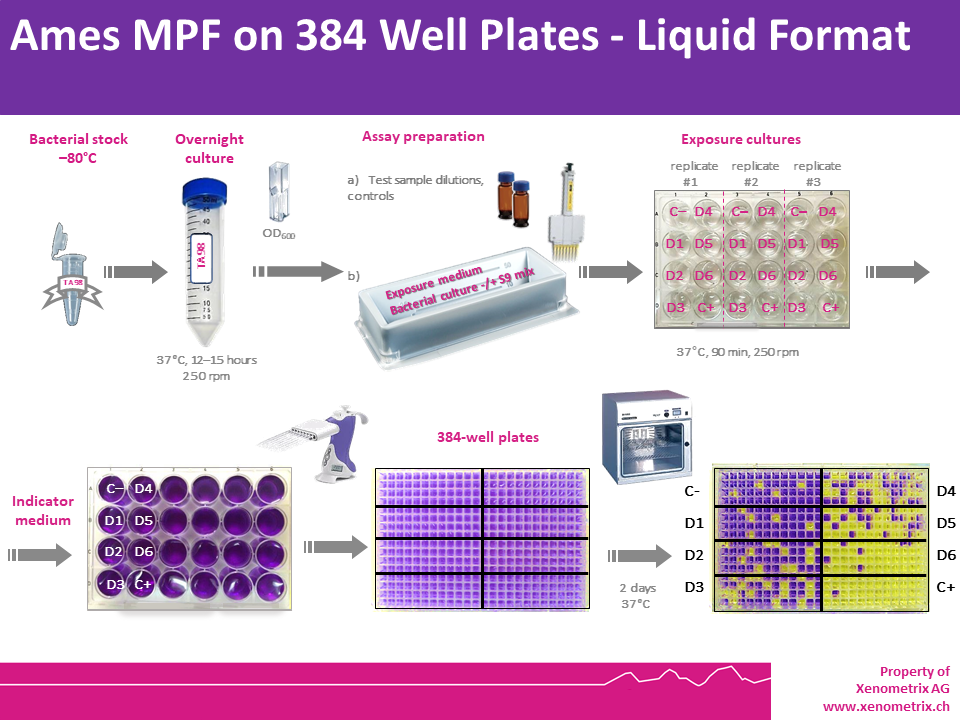
Bacteria are exposed to 6 concentrations of a test sample, as well as a positive and a negative control, for 90 minutes in medium containing enough histidine (S. typhimurium) or tryptophan (E. coli) to support approximately two cell divisions. After exposure, the cultures in each condition (negative control, test samples and positive controls) are diluted in pH indicator medium lacking histidine or tryptophan and aliquoted into 48 wells of a 384-well plate.
Within two days, cells that have undergone reversion to amino acid prototrophy will grow. In the liquid Ames II/Ames MPF™ system, bacterial metabolism reduces the pH of the medium, changing the colour of the well the bacteria are in. The number of wells containing revertant colonies are counted for each dose and compared to a solvent (negative) control. Each dose is tested in triplicate to allow for statistical analysis of the data.
A dose-dependent and significant increase in the number of revertant colonies upon exposure to test sample relative to the solvent controls indicates that the sample is mutagenic.
The mutagenic potential of samples is assessed directly, the compounds are tested in the presence of metabolic activation, provided by a liver homogenate, S9.
Due to the dilution of test compounds in this assay system, coloured compounds can be tested without problems. Volatile compounds are identified in the liquid assay system easily and can be tested with plates sealed with a foil. Also cytotoxic compounds can be detected resulting in a clear purple color due to lysis of bacteria, due to the presence of bubles or due to decreasing number or revertants with increasing compound concentrations
More information on Ames MPF™ Penta 2 – Ames test in 384 well plates, liquid format
More information on Ames MPF™ 98/100 – 2-strain Ames test in 384 well plates, liquid format
Miniaturized Ames Tester Strains – Genotypes
E. coli Ames tester strains as well as S. typhimurium Ames tester strains have been used for more than 40 years to detect mutagenic compounds in chemicals, pharmaceuticals, copsmetics, biocides, water and other environmental samples. They are all listed in the guideline OECD 471: Bacterial Reverse Mutation Test and in guideline ICH M7(R1) for genotoxic impurities. Point mutations were made in the histidine (Salmonella typhimurium) or the tryptophan (Escherichia coli) operon, rendering the bacteria incapable of producing the corresponding amino acid. These mutations result in his- or trp- organisms that cannot grow unless histidine or tryptophan is supplied.
The mutagenic potential pf a test sample is assessed by exposing these amino acid-requiring organisms to varying concentrations of sample and selecting for the reversion event. Media lacking histidine or tryptophan are used for this selection which allow only those cells that have undergone the reversion to histidine / tryptophan prototrophy to survive and grow. A mutagenic event causing base substitutions or frameshift mutations within the gene may cause a reversion to amino acid prototrophy. These reverted bacteria will then grow in histidine- or tryptophan-deficient media, respectively, whereas non reverted bacteria will not be able to grow. The media during the growth phase can be liquid or agar based
E. coli WP2 strains are used in agar based Ames test, in miniaturized agar based (MicroAmes24) and liquid format Ames test (Ames MPF™). The strains are used in accordance with Guidelines OECD TG 471 and ICH M7(R1).
More information on Ames Tester Strains.
Ultraminiaturized NanoAmes™ – Assay Description
Bacteria are exposed to different and ultralow concentrations (< 45 µg) of a test sample or a mixture, as well as a positive and a negative control in a medium containing limited quantities of histidine (S. typhimurium) or tryptophan (E. coli) to support approximately two cell divisions. After exposure, the cultures in each condition (negative control, test samples and positive controls) are poured on an agar support in multiwell plates (25-well plates). Reducing the surface area (comparatively to a regular 12 cm Petri dish) and adjusting the protocol, the amount of test sample needed to run the test is dramatically reduced, while keeping similar limits of detection.
Within three days, cells that have undergone reversion to amino acid prototrophy will grow and form colonies. In the ultraminiaturized Ames format NanoAmes™ (Service Analytics available, kit in development) and NanoAmes MPF™ (kit in development), revertant bacteria will form colonies, whose counts will be compared to those grown in the solvent (negative) control wells. Each dose is tested in quintuplicates to allow for statistical analysis of the data.
A dose-dependent and significant increase in the number of revertant colonies upon exposure to test sample relative to the solvent controls indicates that the sample is mutagenic.
The mutagenic potential of samples is assessed directly and in the presence of metabolic activation, provided by a liver homogenate, S9.
More information on NanoAmes™ – Ultra-Miniaturized 25-well agar plate Ames test – in development.
Miniaturized Ames test – References
- Brooks TM. 1995. The use of a streamlined bacterial mutagenicity assay, the MINISCREEN. Mutagenesis. 10(5):447–8.
- Burke DA, Wedd DJ, Burlinson B. 1996. Use of the Miniscreen assay to screen novel compounds for bacterial mutagenicity in the pharmaceutical industry. Mutagenesis. 11(2):201–5.
- Flamand N, Meunier J, Meunier P, Agapakis-Caussé C. 2001. Mini mutagenicity test: A miniaturized version of the Ames test used in a prescreening assay for point mutagenesis assessment. Toxicol In Vitro. 15(2):105–14.
- Cariou O, et al. in preparation.

 Navigation
Navigation